With over 30 million diabetics in the U.S. – a number that grows larger and larger each day – patients suffering from type 2 diabetes are always looking for new solutions and novel treatments to help with their condition. Similarly, pharmaceutical companies are searching for new approaches to treating diabetes and lessening its symptoms. When sodium-glucose cotransporter 2 (SGLT2) inhibitors first hit the market in 2013, it looked like they might change the landscape of how type 2 diabetes was treated.
Unfortunately, in just 3 short years, a growing tide of reported problems is amassing against these types of diabetes drugs, the latest of which involves Invokana® (canagliflozin) – the class’s top-selling drug, manufactured by Janssen Pharmaceuticals – and Invokamet™, which is typically taken in combination with Metformin®. The 2 drugs share the same active ingredient: canagliflozin.
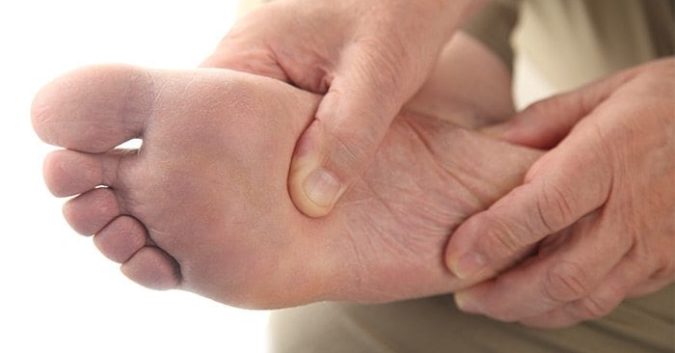
Just last Friday, May 22nd, the U.S. Food and Drug Administration (FDA) alerted the public of new interim safety results that show type-2 diabetics taking canagliflozin may also bear a doubled risk of leg and foot amputation, too.
Invokana and other SGLT2 inhibitors such as Farxiga®, Glyxambi®, and Jardiance® are not strangers to controversy. In fact, they have been the center of numerous public health notices, consumer warnings, and alleged marketing controversies. Over 100 lawsuits have been filed over Invokana alone – many of which involve the dangerous blood condition ketoacidosis.
An Even Newer Warning for the Newer Class of Drug
In the recent Canagliflozin Cardiovascular Assessment Study (CANVAS) trial, which tracked 4,500 type-2 diabetics for an average of 4.5 years, results yielded data showing amputations occurred twice as often in patients taking canagliflozin compared against the control group, who were given a placebo.
The study’s interim analysis found that the risks of amputation were the equivalent to 7 out of every 1,000 patients who took 100mg of canagliflozin daily and 5 out of every 1,000 patients taking 300mg of canagliflozin daily. Those taking a placebo in the trials had a lower risk for amputation, at the rate of 3 out of every 1,000 patients.
The FDA has been quick to note that this is not a definitive cause and effect; at this time, more studies will need to be conducted, and the recent findings only indicate what is considered to be a correlation.
An Older, Persistent Problem: Acid in the Blood
While leg and foot amputations are making recent headlines, one of the drug category’s most consistent problems has been its link to the dangerous blood condition called ketoacidosis. This condition emerges when a higher-than-normal level of blood acids called ketones build up in the blood. Ultimately, ketoacidosis may lead to diabetic coma or even death.
The symptoms of ketoacidosis include excessive thirst, frequent urination, difficulty breathing, nausea, vomiting, abdominal pain, confusion, and unusual fatigue or sleepiness.
Invokana works by lowering the blood sugar level in type 2 diabetes patients by encouraging the body to filter out excess glucose from the blood and excrete it through urine. This excretion prevents glucose from reentering the kidneys. This process, however – unique to SGLT2 inhibitors – has its apparent consequences. The drug class has also been linked to kidney and bladder cancer, as well as an increased risk for heart attack and stroke.
It was just last year that the FDA upped Invokana’s warning label to include an additional warning for ketoacidosis, or “acid in the blood”; they also warned of potentially serious urinary tract infections. Earlier this year, the European Medicines Agency (EMA) followed suit.
Moving forward as a User of an SGLT2 Inhibitor: What to Know
Ketoacidosis is a serious problem, and it must be acted upon immediately in order to prevent irreversible harm or death.
In regards to the newer FDA alert regarding amputation, patients are being warned to seek immediate medical attention if they feel any pain, tenderness, sores, or ulcers in their legs or feet.
Diabetics are already predisposed for elevated risks of leg, foot, and toe amputation; so it is important that patients speak to their doctors about any and all symptoms they experience. Meanwhile, the FDA will continue to investigate the correlation, and will update the public with more information as it becomes available.